Shanghai, July 10, 2025 — Researchers led by Professor Minghui Zhu from East China University of Science and Technology (ECUST) have made a significant advance in understanding hydrogen production catalysts. By developing an innovative dynamic spectroscopic technique, the team has, for the first time, deciphered the real-time reaction network of methanol steam reforming (MSR), clarifying the bifunctional catalytic mechanism at the copper-zinc (Cu-Zn) interface. This work provides a crucial theoretical foundation for the rational design of high-performance hydrogen production catalysts and is published in the Journal of the American Chemical Society.
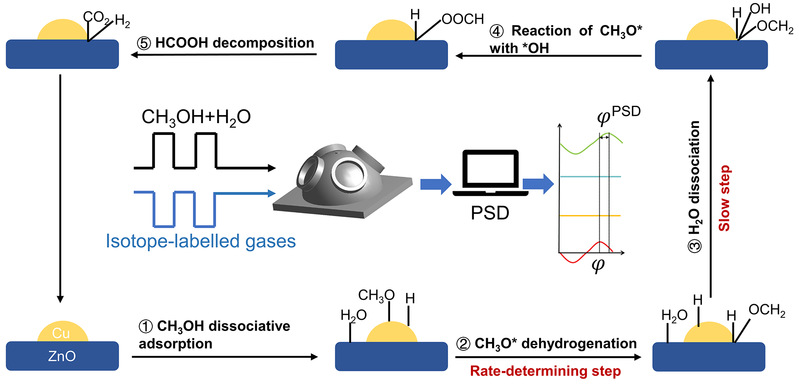
A clear understanding of the active sites and mechanism in MSR, a promising hydrogen generation technology, has been limited by static experimental methods. To overcome this, the team pioneered an isotopic Modulated Excitation Phase-Sensitive Detection DRIFTS (ME-PSD-DRIFTS) method. This approach dynamically tracks active intermediates on the catalyst surface by periodically switching isotopic feedstocks (like H/D and ¹²C/¹³C), effectively distinguishing them from inactive spectator species.
The study revealed the decisive synergistic role of the Cu-Zn interface: methanol preferentially adsorbs and dissociates on Cu sites to form methoxy (CH₃O), while ZnO primarily adsorbs water. A key finding is that the formate intermediate (HCOO) forms via interaction between methoxy and surface hydroxyls, with its hydrogen originating from methanol, not water. The phase-sensitive detection further identified the dehydrogenation of methoxy as the rate-determining step, confirming that formate directly decomposes into CO₂ and H₂, ruling out a CO pathway.
These insights resolve long-standing controversies about the active sites and mechanism in MSR over Cu/ZnO catalysts, paving a new way for studying complex reaction mechanisms in heterogeneous catalysis.