Tuning Catalyst Microenvironments Breaks Selectivity Barrier for Key Chemical Production
March 21, 2025 — Researchers from East China University of Science and Technology (ECUST) have developed a novel catalyst design strategy that overcomes a critical challenge in producing polymer-grade propylene. A team led by Professor Xuezhi Duan, Special Researcher Yueqiang Cao, and Professor Sheng Dai has created bimetallic catalysts with precisely tuned active sites, achieving both high activity and exceptional selectivity in propyne semi-hydrogenation. The work is published in Angewandte Chemie International Edition.
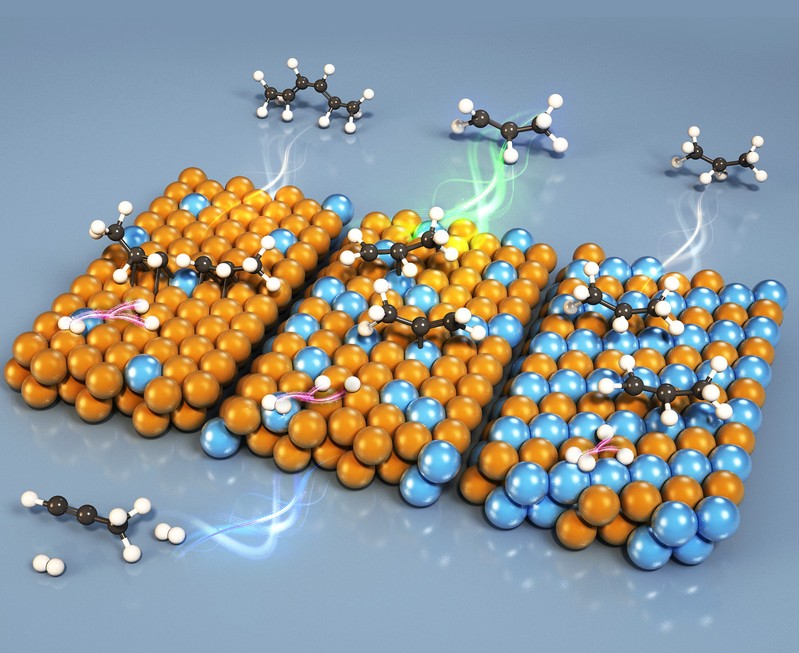
The selective hydrogenation of propyne to propylene is vital in the chemical industry. However, traditional copper (Cu) catalysts suffer from weak hydrogen activation, leading to side reactions, while palladium (Pd)-based catalysts often cause over-hydrogenation due to overly strong propylene adsorption.
The team solved this by constructing catalysts with different local environments by precisely controlling the Pd/Cu ratio. They found that Pd cluster sites on a Cu surface were optimal. These sites not only directly catalyze the hydrogenation reaction but also promote hydrogen dissociation and, crucially, the desorption of the desired propylene product. This dual function breaks the traditional activity-selectivity trade-off.
Performance tests confirmed the breakthrough: the optimized catalyst achieved complete propyne conversion with a propylene selectivity of 95.3%. In contrast, catalysts with Pd single atoms or a PdCu alloy structure were more prone to side reactions.
This research provides a new principle for the rational design of advanced hydrogenation catalysts by atomically tailoring the local environment of active sites to control reaction pathways.