Shanghai, November 12, 2025 — Researchers from the School of Chemical Engineering at East China University of Science and Technology (ECUST) have achieved a significant advance in catalytic hydrogenation technology. Their work on a novel inverse NiOx–Ag interface strategy successfully decouples the activation of reactant molecules, overcoming the long-standing challenge of balancing activity and selectivity in ester hydrogenation reactions. The findings have been published in the prestigious journal Angewandte Chemie International Edition.
Ester hydrogenation is a critical process in producing chemicals for polyesters, biodegradable plastics, fine chemicals, and fuel additives. A central problem has been that both the inert hydrogen molecule (H₂) and the polar carbonyl group in esters typically compete for activation on the same metal sites in conventional catalysts. This competition limits reaction efficiency and often leads to unwanted side products like over-hydrogenation.
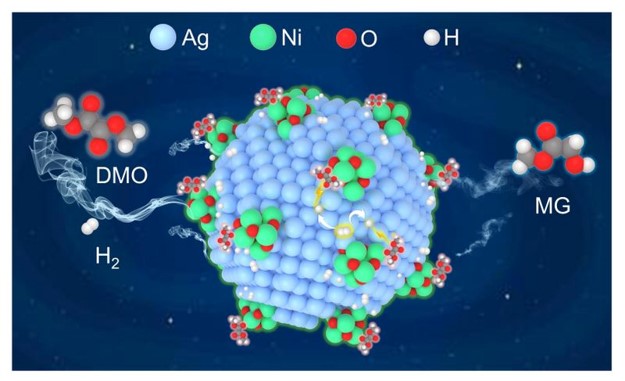
To solve this, the ECUST team developed an inverse NiOx-Ag interface structure. They constructed a catalyst where nickel oxide (NiOx) species are distributed over a silver (Ag) surface. Advanced characterizations confirmed the creation of this unique interface with electron-deficient NiOx and electron-rich Ag sites.
This design spatially separates the activation processes: ester molecules adsorb and activate on the NiOx sites, while H₂ molecules dissociate on the adjacent Ag sites. Hydrogen atoms then efficiently spill over to the reaction site. This decoupling mechanism was verified by in-situ spectroscopy and density functional theory calculations, which showed reduced energy barriers for the key steps.
The catalyst, tested for dimethyl oxalate (DMO) hydrogenation to methyl glycolate (MG), demonstrated exceptional performance. It achieved near-complete DMO conversion (接近100%) and 99% selectivity toward MG under mild conditions, and maintained stable operation for over 500 hours—surpassing all previously reported catalysts.
The research provides a new, rational design principle for creating efficient interfacial catalysts for complex hydrogenation reactions and related industrial processes.